Infrared Radiation and Kinetic Theory- Recap infrared radiation
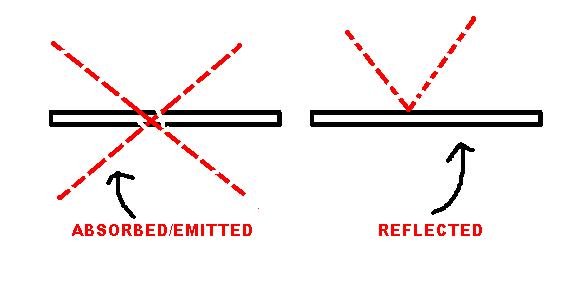
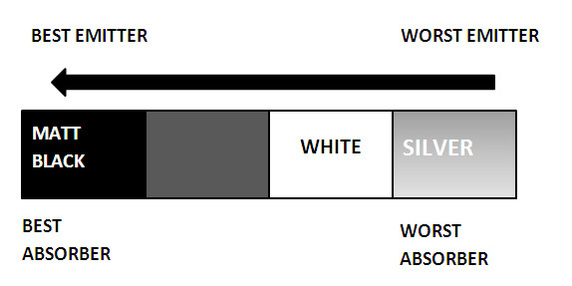
- Use the kinetic theory to explain different states of matter - Describe the differences in energy of the particles in solids, liquids and gases. DIAGRAMS What is each surface likely to be made of? Thinking caps on please. From the diagram ABOVE you should be able to recognise which surface would be of which material and colour. No? The first one labelled 'Absorbed/Emitted' would be a matt black surface. These surfaces are the best emitters and best at absorbing heat energy. However, the surface which 'reflected' the ray of heat is likely to be a shiny silver surface. This means most heat energy is reflected and not much is absorbed or emitted. |
Infrared radiation is one method in which heat energy can be transferred. It can travel through a vacuum and can be absorbed and reflected. The diagram below shows the different types of material which do different things when it comes to emitting and absorbing heat energy.
Something to think about ...
|
ALL OF THE ABOVE HAS NOTHING TO DO WITH PARTICLES BUT WITH INFRARED RADIATION.
Juuuuuuuuuuuuuuust checking ....
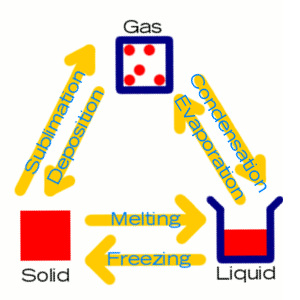
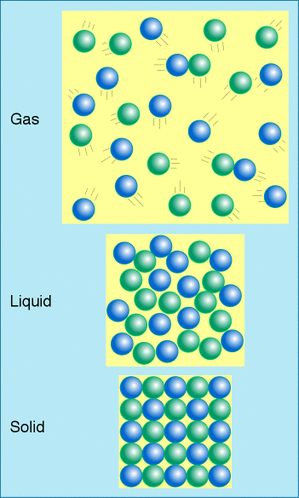
Kinetic TheoryTHE KINETIC THEORY. Gosh, reminds you of the wonderous years in KS3 doesn't it? Well, if you're sitting there thinking 'Ha ha ha, i have no clue. SHINY OBJECT' then have no fear. All shall become clear (WOAH Shakespeare in the making). Solids, liquids and gases. If you look at the diagram on the right if displays the whole cycle. Solids - tightly packed structure, forces of attraction (wink) are strong, not much vibration, high density and not much kinetic energy. Liquids - Loosely packed, forces of attraction are medium, medium density, slight movement and little kinetic energy. Gases - Freely packed, weak forces of attraction, lots of movement, low density and high kinetic energy. Condensing causes HEATING because the energy released when new bonds are made between the gas particles goes into the liquid. Evaporating causes COOLING because the energy needed to break bonds between particles for them to evaporate is taken from the liquid. In the exam they may ask you something like this ...
|
TIP!Most of us, if we see a question telling us to draw solids liquids and gases, would think to ourselves 'pfffft primary schools stuff I don't need to think'. Word of wisdom, don't think that. The examiners are VERY fussy when it comes to drawing these. Make sure the atoms in your solid are all the same size, shape and aligned straight. Make sure your liquids have a bit of room, same size and shape. Make sure you have only a few atoms on your gases state diagram. Make sure over a half of the diagram is empty when drawing the gas atoms and that they are still the same shape and size atoms. NEXT STAGE |